Overview
Citrulline is a non-proteinogenic amino acid that is produced through post-translational deimination of peptidyl-arginine. Peptidyl-arginine deiminases catalyze the hydrolysis of a guanido group into an urea group. This modifications affects the formation of hydrogen bonds and therefore protein folding.
| pKa | NC | Loss | Gain | Deltamass | H | AA | UV-Spec | Pattern |
| – | No | NH | O | Av: 0.9848 M: 0.9840 | – | R | – | – |
In-depth mechanism
Citrulline is a non-proteinogenic amino acid that is produced through post-translational deimination of peptidyl-arginine. Peptidyl-arginine deiminases (PADs) catalyse the hydrolysis of a guanidogroup into an urea group. PADs are calcium dependent enzymes1. This process is called citrullination or to a lesser extent also known as arginine deimination and is shown in figure 1.
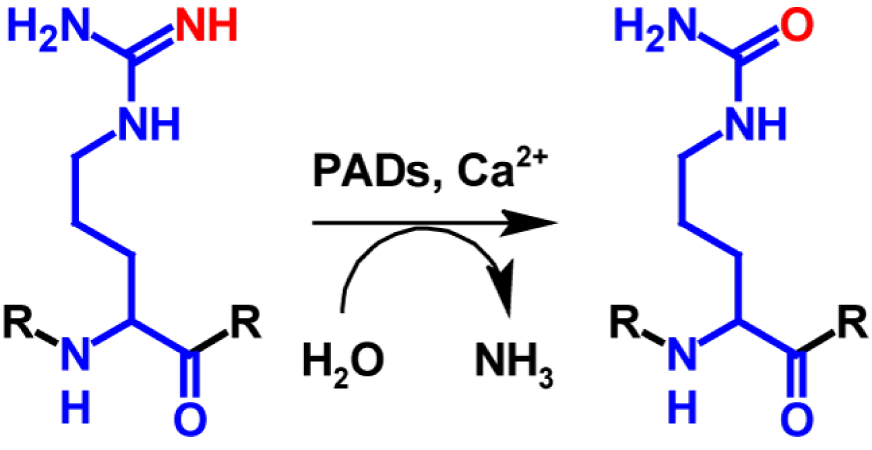
Each Citrullination leads to a mass increase of 0.984 Da. In addition, citrulline is electrically neutral unlike the positively charged arginine, resulting in a loss of a positive charge. This has are markable effect on the acidity of the amino acid side chain. The isoelectric point is lowered from 11.41 for arginine to 5.91 for citrulline because the newly formed urea group is not ionizable unlike the guanido group2. This affects hydrogen bond formation and protein folding, which ultimately results in altered hydrophobicity and protein–protein interactions. In extreme cases it can even lead to denaturation. The exact biological function of citrullination remains unclear. However, it is recognized as central for transcriptional regulation of gene expression. About 10 % of all histones are citrullinated, which underlines the importance of this post-translational modification in many nucleus-associated processes. Citrullination also plays an important role in triggering formation of neutrophil extracellular traps. This is an important defence mechanism against bacterial pathogens1.
References
- 1.Yuzhalin AE. Citrullination in Cancer. Cancer Res. Published online March 20, 2019:1274-1284. doi:10.1158/0008-5472.can-18-2797
- 2.Valesini G, Gerardi MC, Iannuccelli C, Pacucci VA, Pendolino M, Shoenfeld Y. Citrullination and autoimmunity. Autoimmunity Reviews. Published online June 2015:490-497. doi:10.1016/j.autrev.2015.01.013